Step up: Bacterial Lipase to Substitute Pancreatic Lipase For Enzyme Therapy
D. G. Sani, Melbourne
A. INTRODUCTION
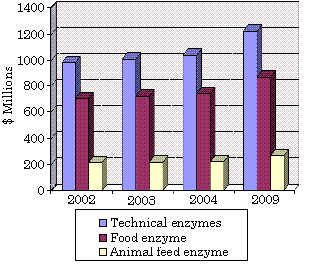
Bacterial lipases have been recognized since nearly 100 years ago in lipase-producing bacteria such as Bacillus pyocyaneus (today is named Pseudomonas aeruginosa), Staphylococcus pyogenes (S. aureus), B. fluorescens (P. fluorescens) [1]. For a lipase to be defined as a true lipase, it must exhibit interfacial activation where a presence of triglyceride should rapidly increase its activity; it should also contain a loop allowing substrate entry to the active site. However, these two criteria are unsuitable for some exceptions where the lipases have the loop but do not exhibit interfacial activation. Hence, a simpler definition of lipases is carboxylesterases that catalyse the hydrolysis and synthesis of long-chain acylglycerols [1]. The prospect for industrial enzymes (technical, food, and animal feed enzyme) is very promising with an estimated increase to $2.4 billion in 2009. Technical enzyme such as in detergents, pulp production, and medical treatment is predicted to have the highest share at 52% [2].
Application of lipase in industrial enzyme production has covered a wide area: flavour development in food technology; as oil removal in detergents; textile making; personal care products e.g. cosmetics; digestive aids for medical treatment [3]. As digestive aids, pancreatic lipase has been widely used in the enzyme therapy to treat lipid malabsorption e.g. in cystic fibrosis and pancreatitis patients. However, there are some barriers with the pancreatic enzyme therapy that prevent its maximal efficacy [3]. Hence, the need of finding an alternative lipase from microorganisms has been raised. Microbial enzymes have several advantages than animals or plants enzymes due to the stability, high yields and rapid production from microbes, and variety in catalytic activity [3]. This paper will firstly describe the background and target market of enzyme therapy for lipase, followed by proposal of methods to isolate lipase-producing bacteria and assays to obtain the acid and protease tolerant lipase.
B. BACKGROUND AND TARGET MARKET
Cystic Fibrosis (CF) is an autosomal genetic disorder affecting mostly Caucasian populations with approximately one in every 3500 newborns is born with CF each year in the United States alone [4]. Being a multi-system disease, CF affects many organs: lungs, pancreas, liver, bones (osteoporosis) [5]. The altered gene in CF disrupts the function of salt transports in organs leading to an excess production of thick, sticky mucus that blocks the ducts in these organs mainly the lungs and the pancreas. Patients with affected pancreas tend to have digestion and growth problems due to pancreatic insufficiency where there is a lack of pancreatic enzyme produced by the body [4,5]. This condition leads to lipid malabsorption because the body cannot break down the essential fatty acid (EFA) molecules coming from external nutrients [6]. Poor nutritional status due to EFA deficiency is correlated with weight and height retardation in children with CF. Moreover, a research by CDC in the US has shown a correlation between nutritional status and the lung function showing increase in body weight in CF patients (measured by BMI) is accompanied by increase in the lung function [4].
Many treatments have been developed to treat the malnutrition in CF patients and improve their life expectancy. To date, pancreatic enzyme (lipase) therapy from porcine origin has been the most widely used since 1930s with about 90% of the patients taking pancreatic enzyme supplements in 2004 [4, 7]. However, the efficiency of this treatment has not reached maximal yet. The pancreatic lipase is found to be denatured by gastric acids before reaching the small intestine where the FA hydrolysis and absorption mainly occur. Although coating the enzyme with an acid resistant (enteric) coating appears to improve the delivery of the enzyme into the small intestine, the low intestinal pH in CF patients after meal (pH less than 4) would still irreversibly denature the pancreatic lipase [7].
Another barrier for the existing pancreatic enzyme therapy is that pancreatic lipase is also denatured by proteases especially chymotrypsin in the intestinal lumen [7]. Hence, in search of ways to improve the enzyme therapy, the aim is to find an alternative lipase that would be acid resistant, protease resistant and would still possess the pancreatic lipase activity.
C. SCIENTIFIC BACKGROUND FOR BACTERIA ISOLATION
In order to find a lipase which shares features with pancreatic lipase, one must know the characteristics of pancreatic lipase. It is known that pancreatic lipase hydrolyses long-chain triacylglyceride (TG) into FAs to be readily absorbed in the small intestine at the optimal pH of 8 [6]. When the pH falls below six the pancreatic lipase is rendered inactive and if the pH less than 4.5, it is irreversibly inactivated; the later is the case in CF patients causing denaturation of the pancreatic lipase in the small intestine during the enzyme treatment [6, 7]. Hence, the alternative lipase would have to be able to survive under acidic environment (pH less than 4). Moreover, knowledge of the active site structures of pancreatic lipase can be a valuable information in finding a new lipase. The active site residues of pancreatic lipase are serine (Ser 152), aspartate (Asp 176), and histidine (His 263) [8]. These are found to be conserved in bacterial lipases where the active site has a nucleophilic residue (Ser, Cys or Asp), a catalytic triad residue (Asp or Glu), and a histidine [1].
Acid resistant lipase could most probably be obtained from bacteria living in acidic environment. A study shows that Salmonella, a gastrointestinal pathogen, produces proteins: RpoS, PhoP, and Fur that help them survive under the harsh gastric environment. RpoS and Fur are responsible for survival against organic (weak) acid stress, while PhoP and RpoS act against inorganic acid (low pH) stress [9]. Therefore, bacteria that produce RpoS, PhoP and Fur would presumably be able to survive under low pH in the gastric environment. A database search from the genbank of NCBI returns 33 bacterial strains that have RpoS, PhoP, and Fur proteins in their genome. Most of these strains are from Salmonella, Pseudomonas, E. coli, Yersinia, Shewanella, and Shigella [10].
Out of these bacterial populations, previous studies have shown that some Pseudomonas strains produce true lipase while both E. coli and Salmonella typhimurium produce esterases [1]. Bacteria surviving from the gastric environment would most probably be present in faeces. Hence, novel lipase-producing bacteria can be screened from faecal specimens and looking specifically for Yersinia, and Shigella as no lipases have been identified from these genera before. These specific strains can be isolated from faecal specimens using selective media. For example, MacConkey agar (MAC) is used to isolate lactose-fermenting, gram negative enteric bacteria while xylose lysine desoxycholate (XLD) agar is specific for isolation of Salmonella and Shigella [11]. Yersinia can be isolated on DYS medium containing peptone, ox-bile, arginine, lysine, arabinose, casein where Yersinia will ferment arabinose and appear as bright red colonies [12].
Another consideration in searching for a new lipase is the specificity of the lipase. Since the aim is to screen for a bacterial lipase that mimics pancreatic lipase, the bacterial lipase should be able to hydrolyse long-chain TGs- the substrate for pancreatic lipase [6]. Although the term ‘long-chain’ TG is not strictly defined, in general, glycerolesters with an acyl chain length more than ten carbon atoms can be considered as lipase substrates e.g. trioleoylglycerol. On the other hand, esterase substrates would normally have an acyl chain length less than 10 carbon atoms e.g. tributyrylglycerol. Most lipases, however, are capable of hydrolysing esterase substrates in addition of the lipase substrates [1]. Hence, screening for the alternative lipase would differentiate between lipase and esterase specificities.
D. ASSAYS
Assays that would be used for screening of an alternative bacterial lipase are assays for lipase production, lipase purification, activity and specificity, pH effects on activity (low pH), and tolerance on protease (chymotrypsin).
Identification of lipase-producing bacteria from the formerly isolated strains can be done by a plate assay where the agar medium contains triolein as the lipase substrate. Lipase production on triolein plates is indicated by orange-red fluorescence of the colonies at 350 nm UV lamp [1]. The lipase-producing colonies can then be purified by inoculating a single colony onto agar plates containing essential compounds for growth. Then the strains would be grown in minimal media containing some essential nutrients for growth and enhance extracellular lipase production. It has been studied that exolipase production in Serratia marcescens can be enhanced by addition of certain polysaccharides as shown in the table below [13].
It can be seen that laminarin exhibits the highest exolipase inducing ability followed by glycogen and pectin B, then hyaluronate. Moreover, the exoprotease production should be kept to minimum so that it would not degrade other essential proteins. Amongst these exolipase enhancers, the most suitable polysaccharide would be pectin B since it has high inducing ability with less exoprotease activity produced compared to laminarin and glycogen. Furthermore, due to interfacial activation of most lipases, addition of long-chain triglyceride such as olive oil might enhance the activity. Hence, pectin B and olive oil can be included in the medium as exolipase enhancers.
There are different assays to test lipase activity in the supernatants namely: the photometric assay and titration method [1]. The photometric assay is done using p-nitrophenylpalmitate (pNPP) as the substrate which is mixed with the supernatant [13]. Lipase activity is then monitored by detecting the hydrolysis of various p-nitrophenolesters of FAs (greater than 10 carbon atoms) into p-nitrophenol at 410 nm. Different p-nitrophenolesters would be separately included in the reaction mixture to test for lipase specificity [14]. The titration method measures lipase activity by recording the amount of NaOH used to maintain pH 8 as the fatty acid is liberated after addition of lipase solution into the supernatants [15].
Assay to screen for acid tolerant lipase can be performed by setting up a reaction mixture mimicking the gastric environment where the pH can be as low as 2.5 to 4. The synthetic gastric fluid has been widely used by mixing pepsin, lysozyme, and bile. Although the synthetic gastric fluid is more convenient to prepare, it has been demonstrated that certain bacteria are more sensitive in an ex vivo porcine gastric fluid than in the synthetic gastric fluid at a same pH of 2.5. This suggests that there are some components which are present only in the porcine stomach and can interfere with bacterial survival [9]. Hence, a more reliable assay for acid tolerant lipase can be done using porcine gastric fluid collected from live pigs instead of the synthetic gastric fluid. The pH of porcine gastric fluid can be adjusted from 3.47 to 4.15 by mixing supernatants of several stomach contents with pH ranging from 1.42 to 4.44 [9]. This mixture (synthetic or porcine gastric fluid) can then be added into the bacterial supernatants and then assayed for lipase activity (photometric assay). The acid resistant lipase would retain its ability to hydrolyse p-nitrophenylesters after treatment with the gastric fluid.
Protease tolerant lipase can be assayed by adding a specific protease into the bacterial supernatants prior to the lipase activity assay. For this experiment, chymotrypsin would be used since it is found to denature pancreatic lipase in the small intestine [7]. If successful, the bacterial lipase that retains its activity would be the protease tolerant lipase. After confirming the lipase which possesses the specific characteristics, purification of the lipase can be done by chromatographic methods where the lipase is bound to beads in a column and then eluted with corresponding buffer. For instance, extracellular lipase of Pseudomonas was purified by anion-exchange chromatography and HIC [15]. The main advantage is that purified enzymes have high activity and can be re-used in other reactions.
CONCLUSION
Technical enzymes application in various biotechnology areas has shared the highest part in the global enzymes market since 2002 [2]. One of the major areas is in medical application where pancreatic lipase therapy is used as digestive aids mostly in CF patients. However, the efficacy has not reached maximal due to degradation of the lipase under acidic condition in the stomach and small intestine of these patients [7]. Hence, searching an alternative acid-tolerant lipase from bacterial origin to substitute pancreatic lipase as digestive aids would have high market returns in the future. If successful, the novel bacterial lipase would be acid and protease tolerant where it can retain its activity at pH less than 4. Moreover, purification would increase its activity implying for higher end products yields from fewer lipase mass and hence, effective production cost.
REFERENCES
1. Jaeger, K. E., Dijkstra, B. W., Reetz, M. T. (1999). Bacterial biocatalysts: molecular biology, three-dimensional structures, and biotechnological application of lipases. Annu Rev Microbiol 53: 315-51.
2. BCC report on global enzyme market. http://www.bccresearch.com/bio/BIO030D.asp. Retrieved 5/10/2006.
3. Hasan, F., et. al. (2006). Industrial applications of microbial lipases. http://www.aseanbiotechnology.info/Abstract/21019024.pdf
4. Cystic Fibrosis Foundation. (2004). Patient registry annual data report 2004.
5. Gotz, M. H. (2002). Cystic Fibrosis literature review annual report 2002.
6. Peretti, N., et. al. (2005). Mechanisms of lipid malabsorption in Cystic Fibrosis: the impact of essential fatty acids deficiency. Nutrition & Metabolism 2:11.
7. Durie, P., et. al. (1998). Uses and abuses of enzyme therapy in cystic fibrosis. J R Soc Med 91: 2-13.
8. Olivecrona, G. et al. (1994). “Medical aspects of triglyceride lipases.” In P. Woolley & S.B Petersen (Eds.), Lipases: their structure, biochemistry and application pp.316-318.
9. Bearson, S. M. D., et. al. (2006). Identification of Salmonella enterica serovar Typhimurium genes important for survival in the swine gastric environment. Applied and Environmental Microbiology 72: 2829-36.
10. NCBI web site: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=Genome&cmd=search&term=RpoS,+Fur,+PhoP+containing+bacteria Retrieved 29/9/2006.
11. CDC: Ch4: Identification and isolation of Shigella. http://www.cdc.gov/ncidod/dbmd/diseaseinfo/cholera/ch4.pdf#search='isolation%20of%20shigella'. Retrieved 4/10/2006.
12. Agbonlahor, D. E., et. al. (1982). Differential and selective medium for isolation of Yersinia enterocolitica from stools. J Clin Microbio 15: 599-602.
13. Winkler, U., Stuckmann, M. (1979). Glycogen, hyaluronate, and some other polysaccharides greatly enhance the formation of exolipase by Serratia
marcescens. J Bacteriol 138: 663-670.
14. Kanwar, S. S., et. al. (2006). Purification and properties of a novel extracellular thermotolerant metallolipase of Bacillus coagulans MTCC-6375 isolate. Protein Expression & Purification 46: 421- 428.
15. Kordel, M., et. al. (1991). Extracellular lipase of Pseudomonas sp. strain ATCC 21808: Purification, characterization, crystallization, and preliminary X-Ray diffraction data. J Bacteriol 173: 4836-41.
Summary
Microbial lipases have been more commonly used than lipases derived from plants and animals due to higher stability, rapid production, higher yields and more convenient manipulations of microorganisms. For example, lipase from Mucor miehei is used in cheese production, Candida antartica in surfactants, Serratia marcescens lipase in drugs (diltiazem). Medical area is another promising area where lipase can be used as digestive aids. To date, only pancreatic enzyme (lipase) therapy is used to treat fat malabsorption in Cystic Fibrosis and pancreatitis patients. However, pancreatic lipase is susceptible to low pH (acidic gastric environment) and protease which can render the lipase inactive. Therefore, it is proposed that an alternative bacterial lipase might have the ability to retain its activity under acidic condition and also be protease tolerant. Screenings for this novel bacterial lipase would be done from bacterial populations that are known to survive in the gastric environment. Moreover, different assays would be done to observe the lipase production, activity, and specificity under certain conditions.
A. INTRODUCTION
Bacterial lipases have been recognized since nearly 100 years ago in lipase-producing bacteria such as Bacillus pyocyaneus (today is named Pseudomonas aeruginosa), Staphylococcus pyogenes (S. aureus), B. fluorescens (P. fluorescens) [1]. For a lipase to be defined as a true lipase, it must exhibit interfacial activation where a presence of triglyceride should rapidly increase its activity; it should also contain a loop allowing substrate entry to the active site. However, these two criteria are unsuitable for some exceptions where the lipases have the loop but do not exhibit interfacial activation. Hence, a simpler definition of lipases is carboxylesterases that catalyse the hydrolysis and synthesis of long-chain acylglycerols [1]. The prospect for industrial enzymes (technical, food, and animal feed enzyme) is very promising with an estimated increase to $2.4 billion in 2009. Technical enzyme such as in detergents, pulp production, and medical treatment is predicted to have the highest share at 52% [2].
Global Enzyme Markets Based on Application Sectors, 2002-2009
($ Millions)
($ Millions)
Application of lipase in industrial enzyme production has covered a wide area: flavour development in food technology; as oil removal in detergents; textile making; personal care products e.g. cosmetics; digestive aids for medical treatment [3]. As digestive aids, pancreatic lipase has been widely used in the enzyme therapy to treat lipid malabsorption e.g. in cystic fibrosis and pancreatitis patients. However, there are some barriers with the pancreatic enzyme therapy that prevent its maximal efficacy [3]. Hence, the need of finding an alternative lipase from microorganisms has been raised. Microbial enzymes have several advantages than animals or plants enzymes due to the stability, high yields and rapid production from microbes, and variety in catalytic activity [3]. This paper will firstly describe the background and target market of enzyme therapy for lipase, followed by proposal of methods to isolate lipase-producing bacteria and assays to obtain the acid and protease tolerant lipase.
B. BACKGROUND AND TARGET MARKET
Cystic Fibrosis (CF) is an autosomal genetic disorder affecting mostly Caucasian populations with approximately one in every 3500 newborns is born with CF each year in the United States alone [4]. Being a multi-system disease, CF affects many organs: lungs, pancreas, liver, bones (osteoporosis) [5]. The altered gene in CF disrupts the function of salt transports in organs leading to an excess production of thick, sticky mucus that blocks the ducts in these organs mainly the lungs and the pancreas. Patients with affected pancreas tend to have digestion and growth problems due to pancreatic insufficiency where there is a lack of pancreatic enzyme produced by the body [4,5]. This condition leads to lipid malabsorption because the body cannot break down the essential fatty acid (EFA) molecules coming from external nutrients [6]. Poor nutritional status due to EFA deficiency is correlated with weight and height retardation in children with CF. Moreover, a research by CDC in the US has shown a correlation between nutritional status and the lung function showing increase in body weight in CF patients (measured by BMI) is accompanied by increase in the lung function [4].
Many treatments have been developed to treat the malnutrition in CF patients and improve their life expectancy. To date, pancreatic enzyme (lipase) therapy from porcine origin has been the most widely used since 1930s with about 90% of the patients taking pancreatic enzyme supplements in 2004 [4, 7]. However, the efficiency of this treatment has not reached maximal yet. The pancreatic lipase is found to be denatured by gastric acids before reaching the small intestine where the FA hydrolysis and absorption mainly occur. Although coating the enzyme with an acid resistant (enteric) coating appears to improve the delivery of the enzyme into the small intestine, the low intestinal pH in CF patients after meal (pH less than 4) would still irreversibly denature the pancreatic lipase [7].
Another barrier for the existing pancreatic enzyme therapy is that pancreatic lipase is also denatured by proteases especially chymotrypsin in the intestinal lumen [7]. Hence, in search of ways to improve the enzyme therapy, the aim is to find an alternative lipase that would be acid resistant, protease resistant and would still possess the pancreatic lipase activity.
C. SCIENTIFIC BACKGROUND FOR BACTERIA ISOLATION
In order to find a lipase which shares features with pancreatic lipase, one must know the characteristics of pancreatic lipase. It is known that pancreatic lipase hydrolyses long-chain triacylglyceride (TG) into FAs to be readily absorbed in the small intestine at the optimal pH of 8 [6]. When the pH falls below six the pancreatic lipase is rendered inactive and if the pH less than 4.5, it is irreversibly inactivated; the later is the case in CF patients causing denaturation of the pancreatic lipase in the small intestine during the enzyme treatment [6, 7]. Hence, the alternative lipase would have to be able to survive under acidic environment (pH less than 4). Moreover, knowledge of the active site structures of pancreatic lipase can be a valuable information in finding a new lipase. The active site residues of pancreatic lipase are serine (Ser 152), aspartate (Asp 176), and histidine (His 263) [8]. These are found to be conserved in bacterial lipases where the active site has a nucleophilic residue (Ser, Cys or Asp), a catalytic triad residue (Asp or Glu), and a histidine [1].
Acid resistant lipase could most probably be obtained from bacteria living in acidic environment. A study shows that Salmonella, a gastrointestinal pathogen, produces proteins: RpoS, PhoP, and Fur that help them survive under the harsh gastric environment. RpoS and Fur are responsible for survival against organic (weak) acid stress, while PhoP and RpoS act against inorganic acid (low pH) stress [9]. Therefore, bacteria that produce RpoS, PhoP and Fur would presumably be able to survive under low pH in the gastric environment. A database search from the genbank of NCBI returns 33 bacterial strains that have RpoS, PhoP, and Fur proteins in their genome. Most of these strains are from Salmonella, Pseudomonas, E. coli, Yersinia, Shewanella, and Shigella [10].
Out of these bacterial populations, previous studies have shown that some Pseudomonas strains produce true lipase while both E. coli and Salmonella typhimurium produce esterases [1]. Bacteria surviving from the gastric environment would most probably be present in faeces. Hence, novel lipase-producing bacteria can be screened from faecal specimens and looking specifically for Yersinia, and Shigella as no lipases have been identified from these genera before. These specific strains can be isolated from faecal specimens using selective media. For example, MacConkey agar (MAC) is used to isolate lactose-fermenting, gram negative enteric bacteria while xylose lysine desoxycholate (XLD) agar is specific for isolation of Salmonella and Shigella [11]. Yersinia can be isolated on DYS medium containing peptone, ox-bile, arginine, lysine, arabinose, casein where Yersinia will ferment arabinose and appear as bright red colonies [12].
Another consideration in searching for a new lipase is the specificity of the lipase. Since the aim is to screen for a bacterial lipase that mimics pancreatic lipase, the bacterial lipase should be able to hydrolyse long-chain TGs- the substrate for pancreatic lipase [6]. Although the term ‘long-chain’ TG is not strictly defined, in general, glycerolesters with an acyl chain length more than ten carbon atoms can be considered as lipase substrates e.g. trioleoylglycerol. On the other hand, esterase substrates would normally have an acyl chain length less than 10 carbon atoms e.g. tributyrylglycerol. Most lipases, however, are capable of hydrolysing esterase substrates in addition of the lipase substrates [1]. Hence, screening for the alternative lipase would differentiate between lipase and esterase specificities.
D. ASSAYS
Assays that would be used for screening of an alternative bacterial lipase are assays for lipase production, lipase purification, activity and specificity, pH effects on activity (low pH), and tolerance on protease (chymotrypsin).
Identification of lipase-producing bacteria from the formerly isolated strains can be done by a plate assay where the agar medium contains triolein as the lipase substrate. Lipase production on triolein plates is indicated by orange-red fluorescence of the colonies at 350 nm UV lamp [1]. The lipase-producing colonies can then be purified by inoculating a single colony onto agar plates containing essential compounds for growth. Then the strains would be grown in minimal media containing some essential nutrients for growth and enhance extracellular lipase production. It has been studied that exolipase production in Serratia marcescens can be enhanced by addition of certain polysaccharides as shown in the table below [13].
Table from Winkler, U., Stuckmann, M. (1979). Glycogen, hyaluronate, and some other polysaccharides greatly enhance the formation of exolipase by Serratia
marcescens. J Bacteriol 138: 663-670. Not displayed.
Table Source: Winkler [13].
It can be seen that laminarin exhibits the highest exolipase inducing ability followed by glycogen and pectin B, then hyaluronate. Moreover, the exoprotease production should be kept to minimum so that it would not degrade other essential proteins. Amongst these exolipase enhancers, the most suitable polysaccharide would be pectin B since it has high inducing ability with less exoprotease activity produced compared to laminarin and glycogen. Furthermore, due to interfacial activation of most lipases, addition of long-chain triglyceride such as olive oil might enhance the activity. Hence, pectin B and olive oil can be included in the medium as exolipase enhancers.
There are different assays to test lipase activity in the supernatants namely: the photometric assay and titration method [1]. The photometric assay is done using p-nitrophenylpalmitate (pNPP) as the substrate which is mixed with the supernatant [13]. Lipase activity is then monitored by detecting the hydrolysis of various p-nitrophenolesters of FAs (greater than 10 carbon atoms) into p-nitrophenol at 410 nm. Different p-nitrophenolesters would be separately included in the reaction mixture to test for lipase specificity [14]. The titration method measures lipase activity by recording the amount of NaOH used to maintain pH 8 as the fatty acid is liberated after addition of lipase solution into the supernatants [15].
Assay to screen for acid tolerant lipase can be performed by setting up a reaction mixture mimicking the gastric environment where the pH can be as low as 2.5 to 4. The synthetic gastric fluid has been widely used by mixing pepsin, lysozyme, and bile. Although the synthetic gastric fluid is more convenient to prepare, it has been demonstrated that certain bacteria are more sensitive in an ex vivo porcine gastric fluid than in the synthetic gastric fluid at a same pH of 2.5. This suggests that there are some components which are present only in the porcine stomach and can interfere with bacterial survival [9]. Hence, a more reliable assay for acid tolerant lipase can be done using porcine gastric fluid collected from live pigs instead of the synthetic gastric fluid. The pH of porcine gastric fluid can be adjusted from 3.47 to 4.15 by mixing supernatants of several stomach contents with pH ranging from 1.42 to 4.44 [9]. This mixture (synthetic or porcine gastric fluid) can then be added into the bacterial supernatants and then assayed for lipase activity (photometric assay). The acid resistant lipase would retain its ability to hydrolyse p-nitrophenylesters after treatment with the gastric fluid.
Protease tolerant lipase can be assayed by adding a specific protease into the bacterial supernatants prior to the lipase activity assay. For this experiment, chymotrypsin would be used since it is found to denature pancreatic lipase in the small intestine [7]. If successful, the bacterial lipase that retains its activity would be the protease tolerant lipase. After confirming the lipase which possesses the specific characteristics, purification of the lipase can be done by chromatographic methods where the lipase is bound to beads in a column and then eluted with corresponding buffer. For instance, extracellular lipase of Pseudomonas was purified by anion-exchange chromatography and HIC [15]. The main advantage is that purified enzymes have high activity and can be re-used in other reactions.
CONCLUSION
Technical enzymes application in various biotechnology areas has shared the highest part in the global enzymes market since 2002 [2]. One of the major areas is in medical application where pancreatic lipase therapy is used as digestive aids mostly in CF patients. However, the efficacy has not reached maximal due to degradation of the lipase under acidic condition in the stomach and small intestine of these patients [7]. Hence, searching an alternative acid-tolerant lipase from bacterial origin to substitute pancreatic lipase as digestive aids would have high market returns in the future. If successful, the novel bacterial lipase would be acid and protease tolerant where it can retain its activity at pH less than 4. Moreover, purification would increase its activity implying for higher end products yields from fewer lipase mass and hence, effective production cost.
REFERENCES
1. Jaeger, K. E., Dijkstra, B. W., Reetz, M. T. (1999). Bacterial biocatalysts: molecular biology, three-dimensional structures, and biotechnological application of lipases. Annu Rev Microbiol 53: 315-51.
2. BCC report on global enzyme market. http://www.bccresearch.com/bio/BIO030D.asp. Retrieved 5/10/2006.
3. Hasan, F., et. al. (2006). Industrial applications of microbial lipases. http://www.aseanbiotechnology.info/Abstract/21019024.pdf
4. Cystic Fibrosis Foundation. (2004). Patient registry annual data report 2004.
5. Gotz, M. H. (2002). Cystic Fibrosis literature review annual report 2002.
6. Peretti, N., et. al. (2005). Mechanisms of lipid malabsorption in Cystic Fibrosis: the impact of essential fatty acids deficiency. Nutrition & Metabolism 2:11.
7. Durie, P., et. al. (1998). Uses and abuses of enzyme therapy in cystic fibrosis. J R Soc Med 91: 2-13.
8. Olivecrona, G. et al. (1994). “Medical aspects of triglyceride lipases.” In P. Woolley & S.B Petersen (Eds.), Lipases: their structure, biochemistry and application pp.316-318.
9. Bearson, S. M. D., et. al. (2006). Identification of Salmonella enterica serovar Typhimurium genes important for survival in the swine gastric environment. Applied and Environmental Microbiology 72: 2829-36.
10. NCBI web site: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=Genome&cmd=search&term=RpoS,+Fur,+PhoP+containing+bacteria Retrieved 29/9/2006.
11. CDC: Ch4: Identification and isolation of Shigella. http://www.cdc.gov/ncidod/dbmd/diseaseinfo/cholera/ch4.pdf#search='isolation%20of%20shigella'. Retrieved 4/10/2006.
12. Agbonlahor, D. E., et. al. (1982). Differential and selective medium for isolation of Yersinia enterocolitica from stools. J Clin Microbio 15: 599-602.
13. Winkler, U., Stuckmann, M. (1979). Glycogen, hyaluronate, and some other polysaccharides greatly enhance the formation of exolipase by Serratia
marcescens. J Bacteriol 138: 663-670.
14. Kanwar, S. S., et. al. (2006). Purification and properties of a novel extracellular thermotolerant metallolipase of Bacillus coagulans MTCC-6375 isolate. Protein Expression & Purification 46: 421- 428.
15. Kordel, M., et. al. (1991). Extracellular lipase of Pseudomonas sp. strain ATCC 21808: Purification, characterization, crystallization, and preliminary X-Ray diffraction data. J Bacteriol 173: 4836-41.
Labels: Discovery, enzymes, Innovation

1 Comments:
Thanks a lot for this contribution! It's been very useful for me. Everything is very open and represents very clear explanation of issues. Really blogging is spreading its wings quickly. Your write up is a good example of it. Your website is very useful. Thanks for sharing Bacterial Lipase to Substitute Pancreatic Lipase For Enzyme Therapy.
Purified Enzymes
Post a Comment
<< Home